42 energy diagram for two step reaction
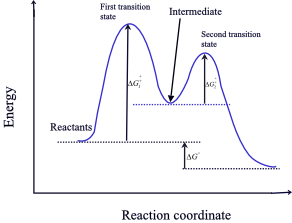
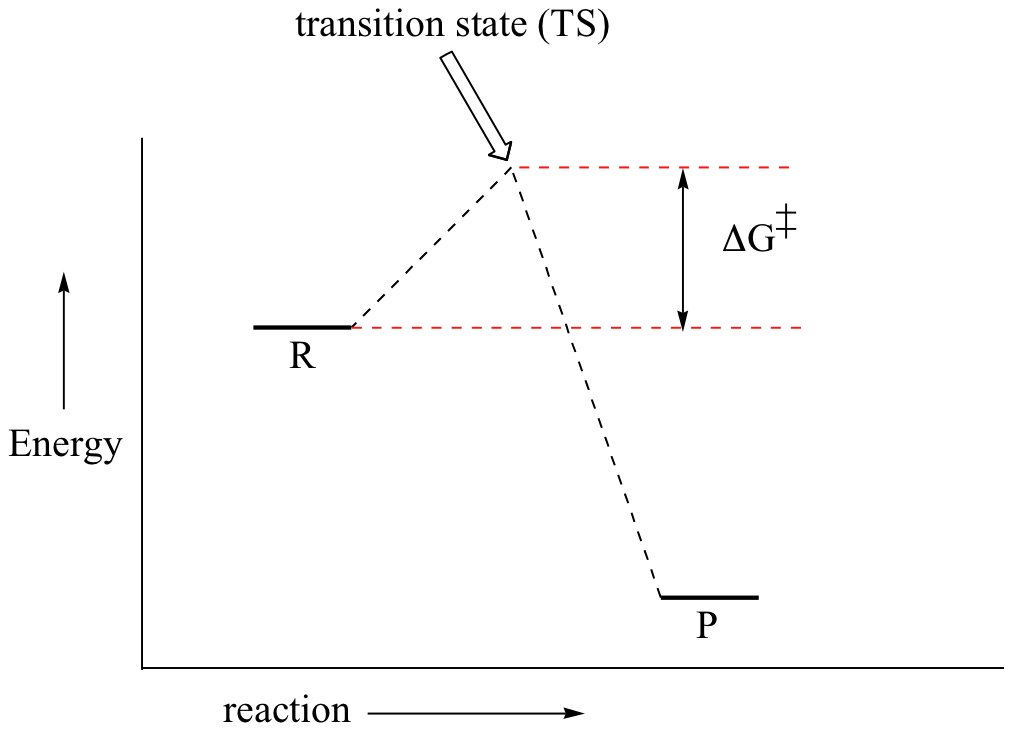
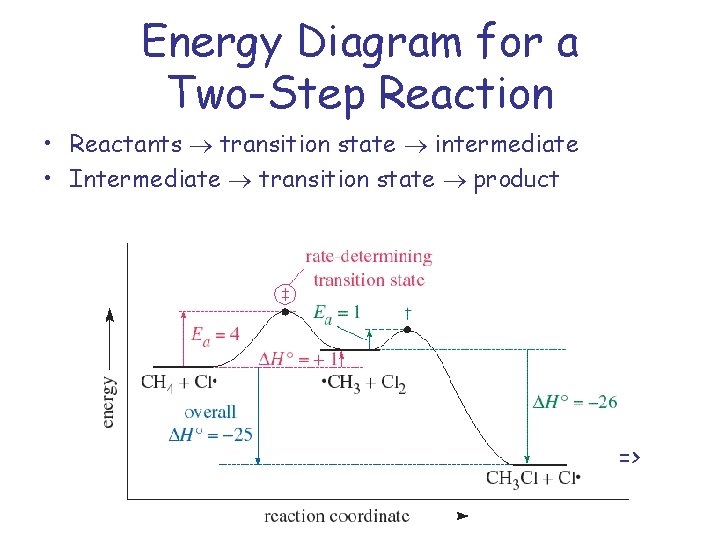
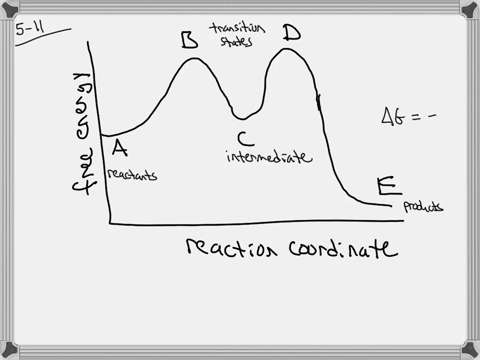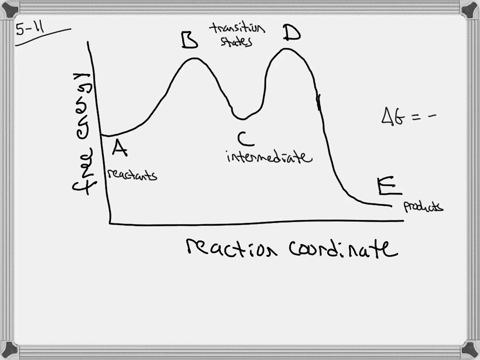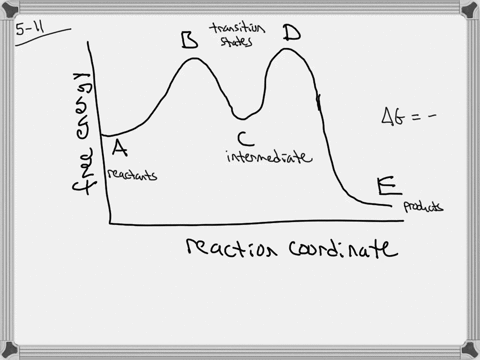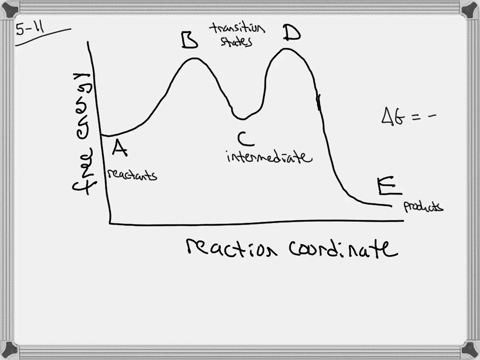
The energy diagram of a two-step reaction is shown below. In the above reaction, a reactant goes through one elementary step with a lower activation energy (transition state 1) to form the intermediate. The intermediate then goes through a second step (transition state 2) with the highest energy barrier to form the product. Draw an energy diagram for a two-step reaction that is exothermic overall, and consists of a fast but endothermic first step, and a slow but exothermic second step. Indicate DG rxn, as well as DG 1 * and DG 2 * for the first and second activation energies, respectively. Label the positions corresponding to the transition states with an asterisk.
Watch Complete videos @ www.LearningChemistryOnline.com Organic Chemistry 1
Energy diagram for two step reaction
• An energy diagram for a two-step reaction with one intermediate. Energy Diagrams. 9 • Organic chemists use a technique called electron pushing, alternatively called arrow pushing, to depict the flow of electrons during a chemical reaction. 57CP. Draw an energy diagram for each reaction. Label the axes, the starting material, product, transition state, ΔH°, and E a. a. A concerted, exothermic reaction with a low energy of activation. b. A one-step endothermic reaction with a high energy of activation. c. A two-step reaction. Label the energy diagram for a two step reaction. Heroin C21h23no5 Pubchem The activation energy for each step is labeled e a1 and e a2. Label the energy diagram for a two step reaction. Endothermic because energy is needed to break the a b bond. Each elementary step has its own activated complex labeled ac 1 and ac 2.
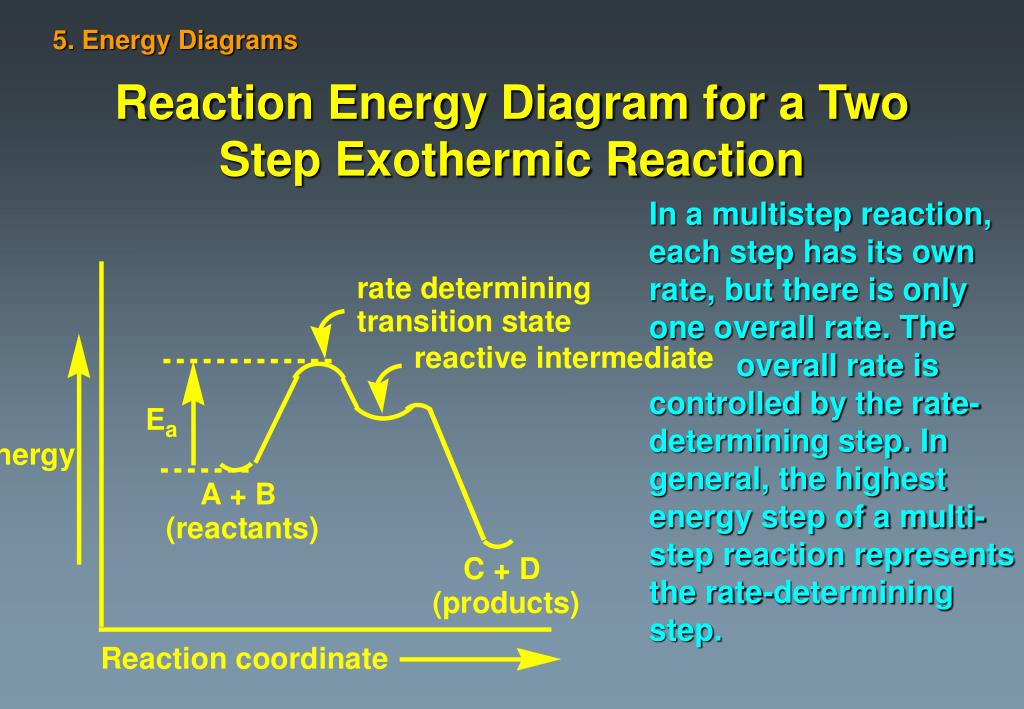
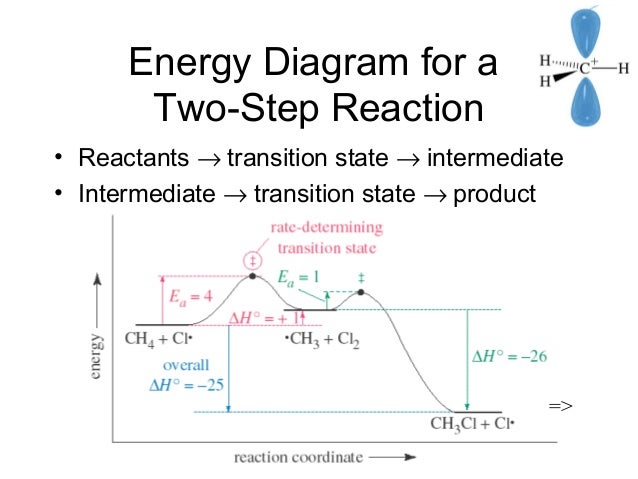
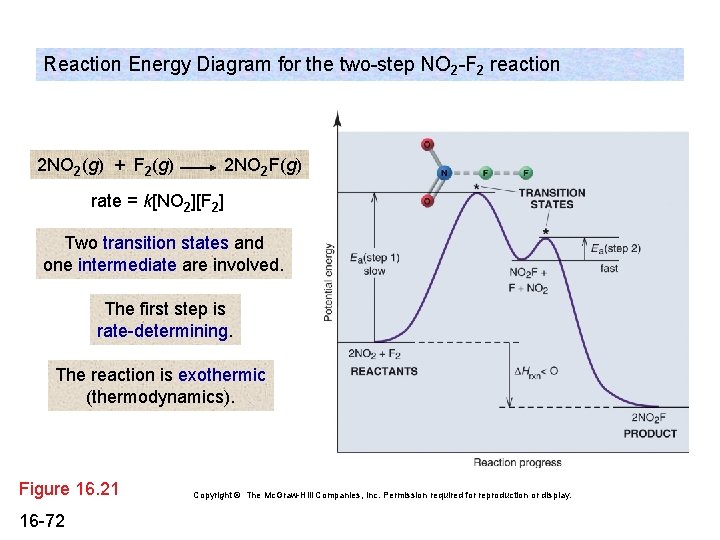
Energy diagram for two step reaction. Energy Diagram for a Two-Step Reaction Involving Formation of an Intermediate Reactive intermediate corresponds to an energy minimum between two transition states Rate-determining step: Step that crosses the highest energy barrier: Slowest step in a multistep reaction Alternate ISBN: 9781305638716, 9781305686465. Organic Chemistry (9th Edition) Edit edition Solutions for Chapter 6 Problem 21E: Draw an energy diagram for a two-step reaction with Keq > 1. Label the overall ΔG°, transition states, and intermediate. Is ΔG° positive or negative? …. The figure above represents the reaction profile of a two step, exothermic reaction. The y-axis represents the potential energy of the reaction species, and the x-axis represents the progress of the reaction. The reaction is exothermic because the energies of the products are lower than those of the reactants. The reactants are represented by the horizontal line at the far left of the graph ... The reaction whose potential energy diagram is shown in the figure is a two-step reaction. The activation energy for each step is labeled E a1 and E a2 .Each elementary step has its own activated complex, labeled AC 1 and AC 2 .Note that the overall enthalpy change of the reaction is unaffected by the individual steps, since it depends only on the initial and final states.
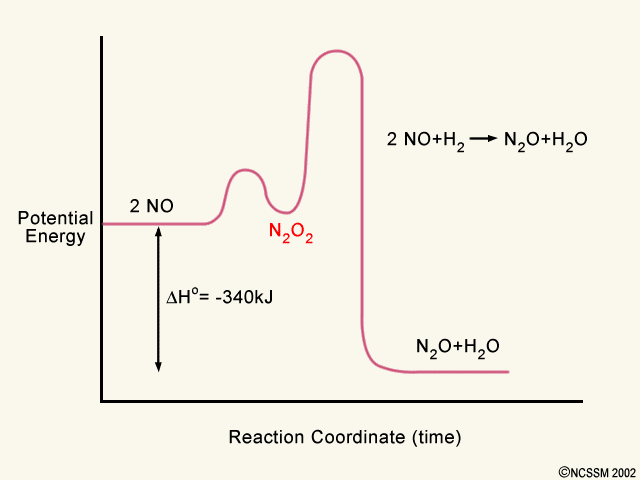
In a similar fashion, two diatomic oxygen molecules are broken apart which requires more energy. Now all of the individual atoms in the reactant molecules have been broken apart. On the right side of the diagram in a second step, the various atoms form new bonds in new molecules of carbon dioxide and water. The activation energy for each step is labeled e a1 and e a2. Label the energy diagram for a two step reaction. It also shows the effect of a catalyst on the forward and reverse activation energy. In the s n 1 reaction the carbocation species is a reaction intermediate. A potential energy diagram for an s n 1 reaction shows that the carbocation ... Draw an energy diagram for a two-step reaction with Keq > 1. Label the overall ΔG°, transition states, and intermediate. Is ΔG° positive or negative? Students also viewed these Organic Chemistry questions. Draw an energy diagram for a one-step reaction with Keq 1. progress of the reaction. The curve should illustrate both the proposed two-step mechanism and the enthalpy change of the reaction. See drawing above. 1 point is earned for the potential energy of the product being lower than the potential energy of the reactants (exothermic reaction). 1 point is earned for a reaction-energy curve that reflects ...
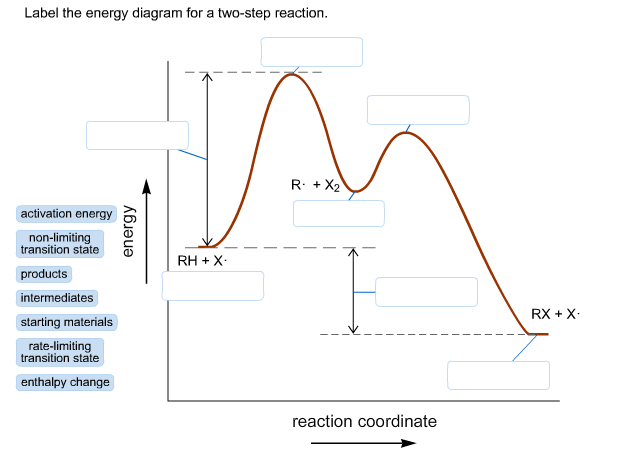
Question. Draw an energy diagram for a two-step reaction, A → B → C, where the relative energy of these compounds is C < A < B, and the conversion of B → C is rate-determining. check_circle. This chemistry video tutorial focuses on potential energy diagrams for endothermic and exothermic reactions. It also shows the effect of a catalyst on the f... This two-step mechanism is illustrated for the reaction of ethene with hydrogen chloride by the following equations. First Step: H 2 C=CH 2 + HCl. H H 2 C=CH 2(+) + Cl (-) Second Step: H H 2 C=CH 2(+) + Cl (-) H H 2 C=CH 2 Cl. An energy diagram for this two-step addition mechanism is shown to the left. Recall that an energy diagram is usually read from left to right. The components of a two-step energy diagram are: • Reactants: are placed on the left/beginning of the energy diagram. • Products: are placed on the right/end of the energy diagram. • Non-limiting transition state: is the transition state with the lowest energy in the energy ...
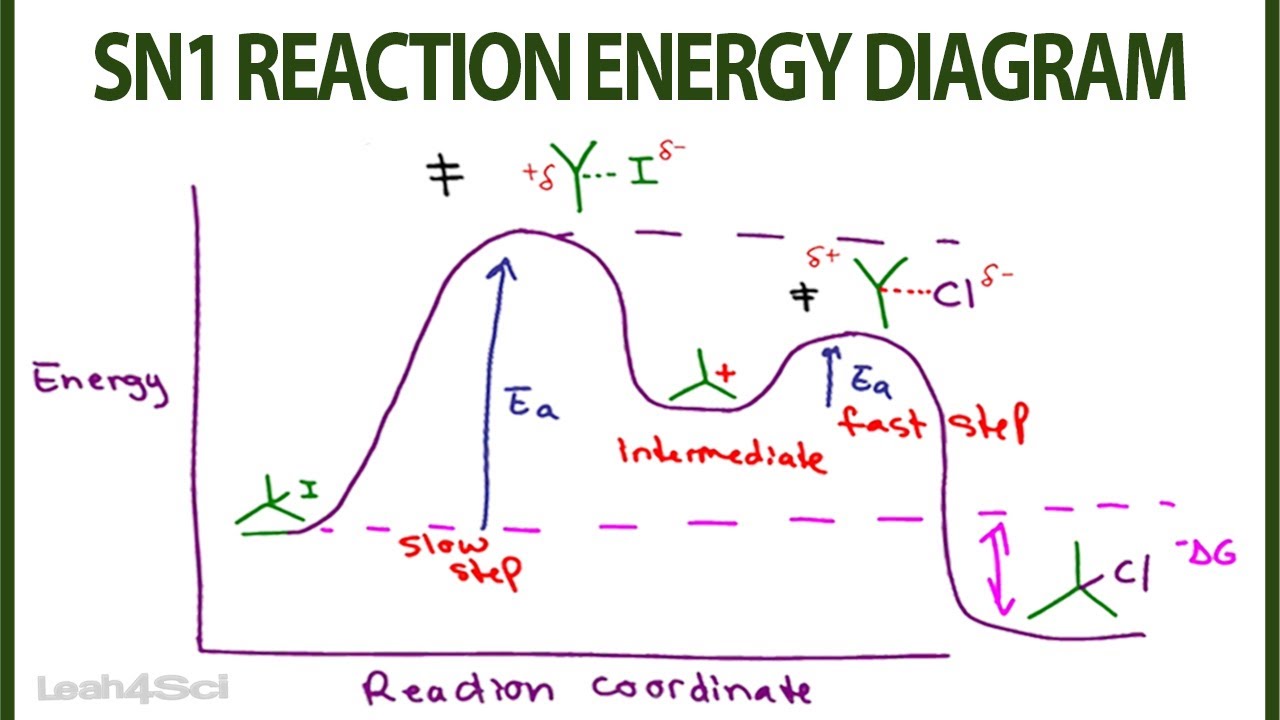
A potential energy diagram for an S N 1 reaction shows that the carbocation intermediate can be visualized as a kind of valley in the path of the reaction, higher in energy than both the reactant and product but lower in energy than the two transition states. Exercise. Draw structures representing TS1 and TS2 in the reaction above.
Jun 10, 2021 · The third step of the citric acid cycle is the first of the four oxidation-reduction reactions in this cycle. Isocitrate is oxidatively decarboxylated to form a five-carbon compound, α-ketoglutarate catalyzed by the enzyme isocitrate dehydrogenase. This reaction, like the second reaction, is a two-step reaction.
Mar 19, 2018 — Label the energy diagram for a two-step reaction. ... An energy profile diagram is a theoretical representation that shows how the energy of the ...
The reaction profile of a two-step reaction is represented by a potential energy diagram. The y-axis depicts the potential energy of the reacting species, while the x-axis depicts the reaction's ...
In the term S N 2, S stands for 'substitution', the subscript N stands for 'nucleophilic', and the number 2 refers to the fact that this is a bimolecular reaction: the overall rate depends on a step in which two separate molecules (the nucleophile and the electrophile) collide. A potential energy diagram for this reaction shows the ...
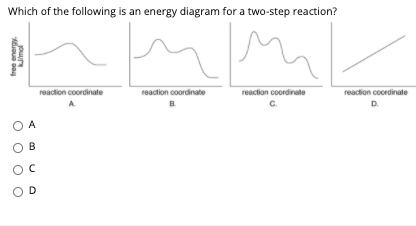
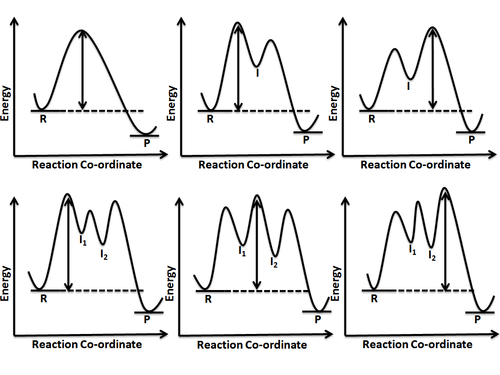
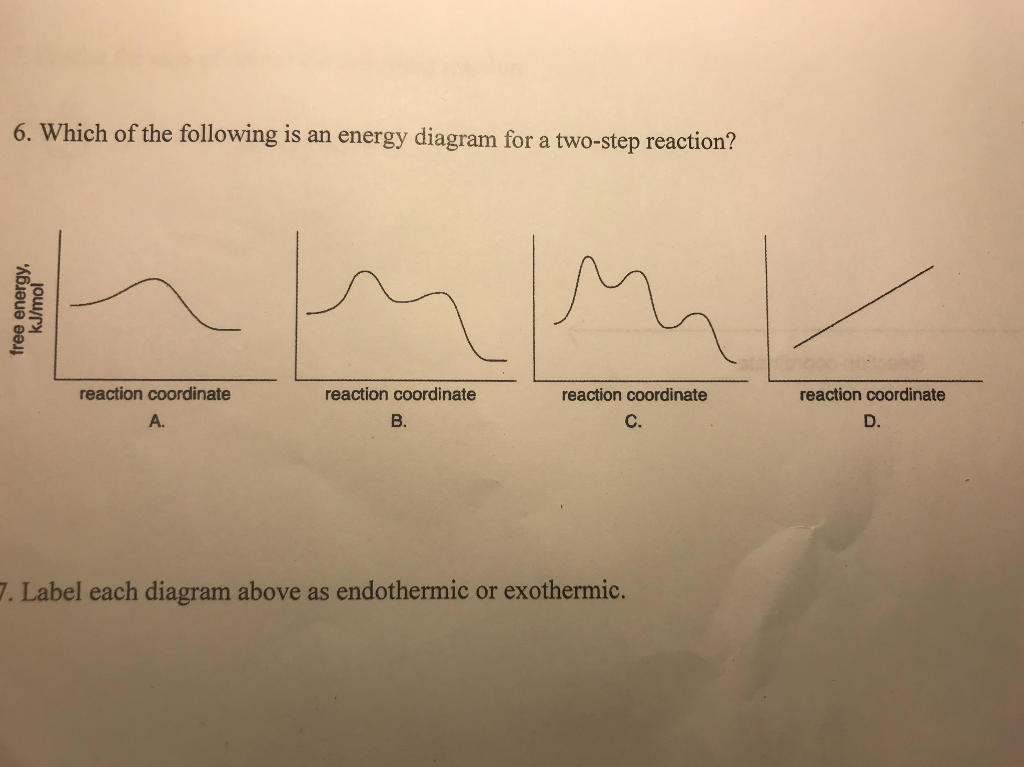
Label the energy diagram for a two-step reaction. Q. A reaction coordinate diagram is shown below for the reaction of A to form E. Answer the following questions.i) Identify the transition state (s)?ii) W... Q. Which reaction coordinate diagram represents a reaction in which the activation energy, Ea, is 50 kj.mol-1 and the ΔHrxn is -15 kj. mol-1?
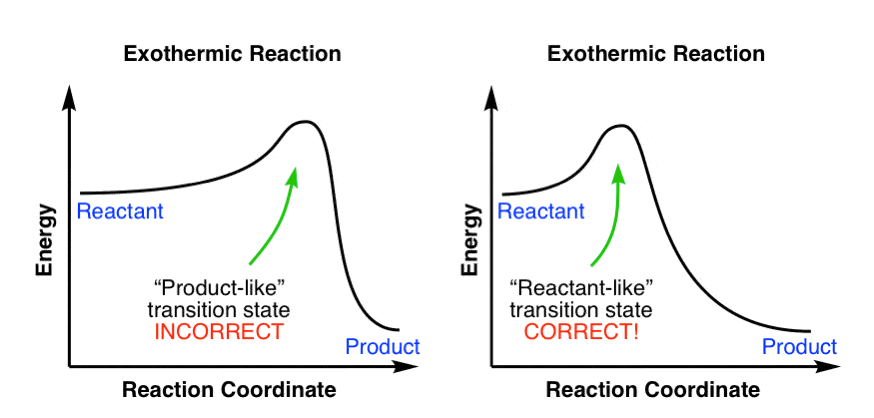
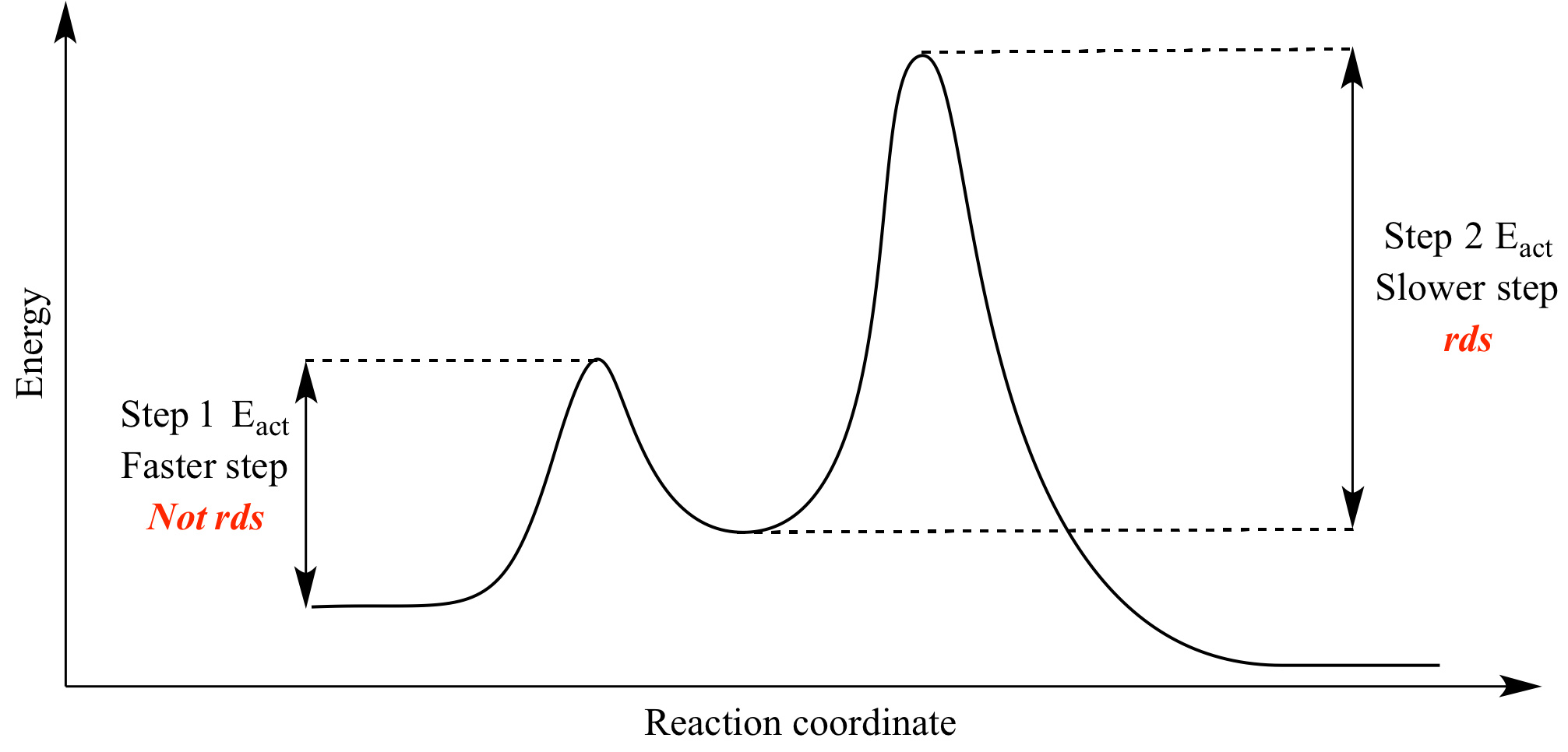
The transition state is the high energy point between two minima along the reaction coordinate. Each step in a mechanism will have a transition state. As with the overall mechanism the rate of the reaction may only depend on the highest energy transition state as this will dominate the rate.
Step off the platform and draw your free body diagram as you fall. You start with no initial velocity, there is no aerodynamic drag, and you are effectively in free fall with an acceleration of 9.8 m/s 2 .
Energy Diagram: The diagram which indicates the number of steps in which the reaction gets completed and the energy required for each step can be shown by plotting graph of rate of reaction versus ...
Nov 01, 2021 · a, Free energy diagram of Ti-OH − adsorption and Ti-OH * creation in the context of the full reaction scheme. Photo-excitation (yellow arrow) separates the diagram into the dark, equilibrated ...
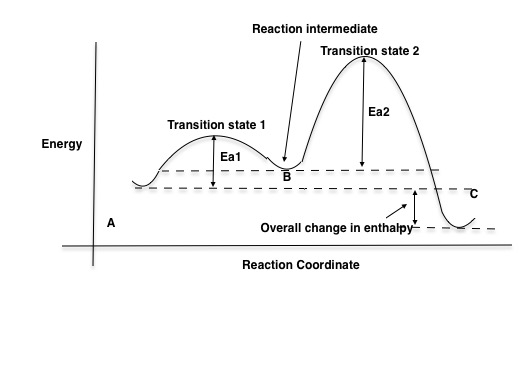
step in a multistep reaction. EXAMPLE 5.1 Draw an energy diagram for a two-step exothermic reaction in which the second step is rate determining. STRATEGY A two-step reaction involves the formation of an intermedi-ate. In order for the reaction to be exothermic, the products must be lower in energy than the reactants. In order for the
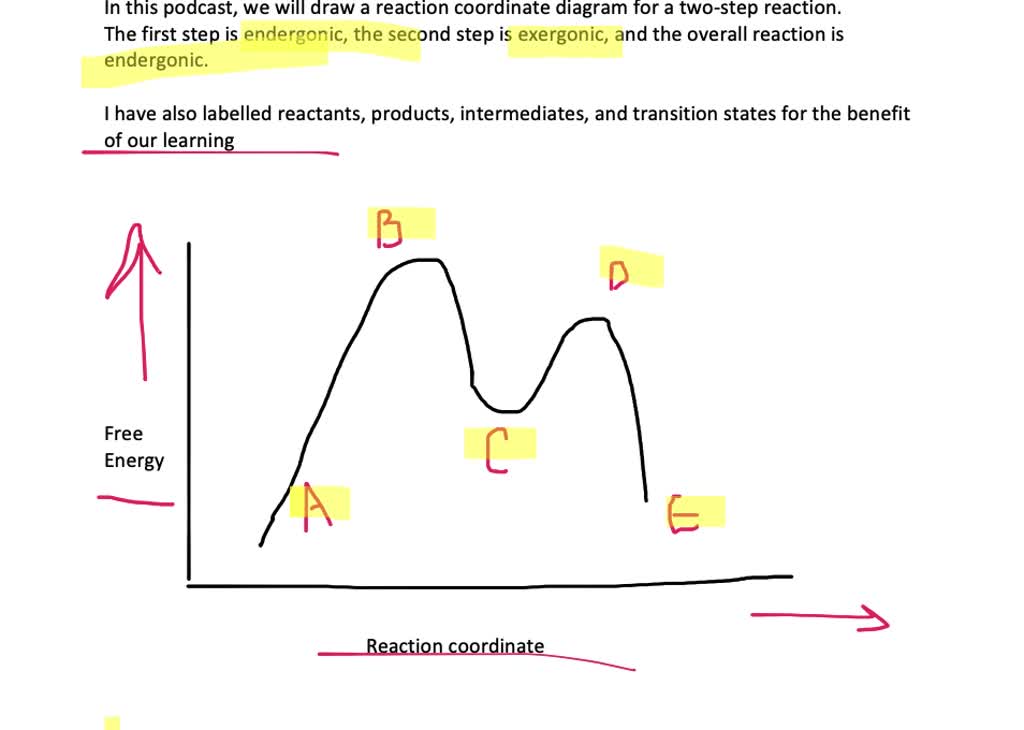
Solved Draw A Reaction Coordinate Diagram For A Two Step Reaction In Which The First Step Is Endergonic The Second Step Is Exergonic And The Overall Reaction Is Endergonic Label The Reactants Products Intermediates
Fusion power is a proposed form of power generation that would generate electricity by using heat from nuclear fusion reactions.In a fusion process, two lighter atomic nuclei combine to form a heavier nucleus, while releasing energy.
Nuclear fusion is a reaction in which two or more atomic nuclei are combined to form one or more different atomic nuclei and subatomic particles (neutrons or protons).The difference in mass between the reactants and products is manifested as either the release or the absorption of energy.
Aug 17, 2018 — The reaction whose potential energy diagram is shown in the figure is a two-step reaction. The activation energy for each step is labeled ...
The reaction coordinate diagram for the ozone photolysis reaction is a little different from those above because this is an endothermic reaction. Together, the products O 2 and atomic O, have a higher energy than the reactant O 3 and energy must be added to the system for this reaction.
d) a spontaneous exothermic reaction. Diagram 2 depending on the values but looks quite small. To be spontaneous needs to be ≤ 20kJ 2. Draw a potential energy (E p ) diagram for a reaction in which ∆H = 80 kJ/mol and
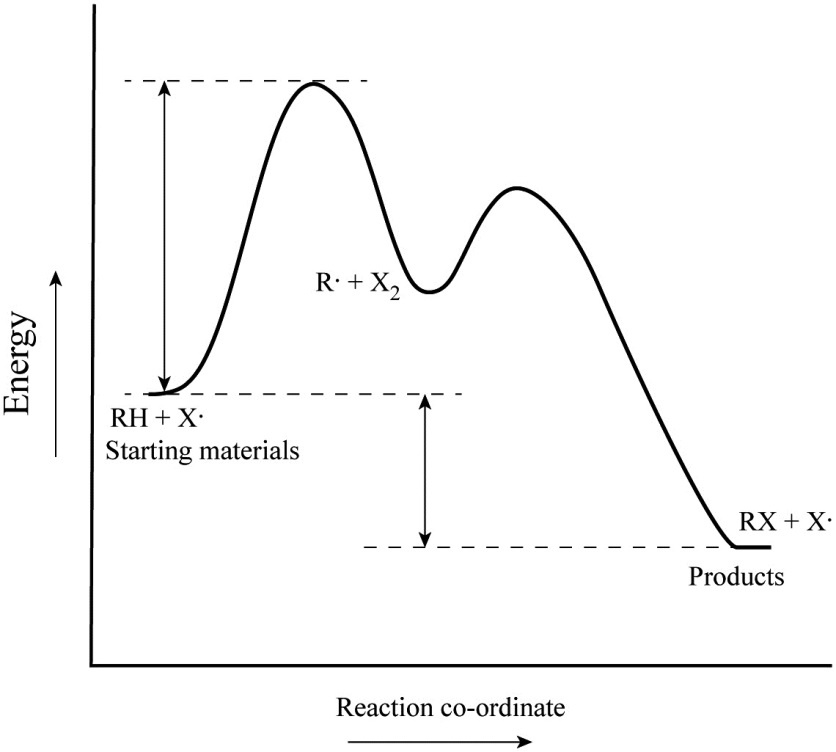
A Two-Step Reaction Mechanism. The transition states are located at energy maxima. The reactive intermediate B+ is located at an energy minimum. Each step has its own delta H and activation energy. The overall energy difference between the starting materials and products is delta H overall. Step 1 has the higher transition energy state, thus it ...
In this reaction, the disulfide F(C 6 H 4)S–S(C 6 H 4)F is formed as a byproduct and removed during a washing step. Although the oxidative removal of the capping agent was conducted for 10–14 days, the presence of residual capping agent was still evident in the EDS spectrum.
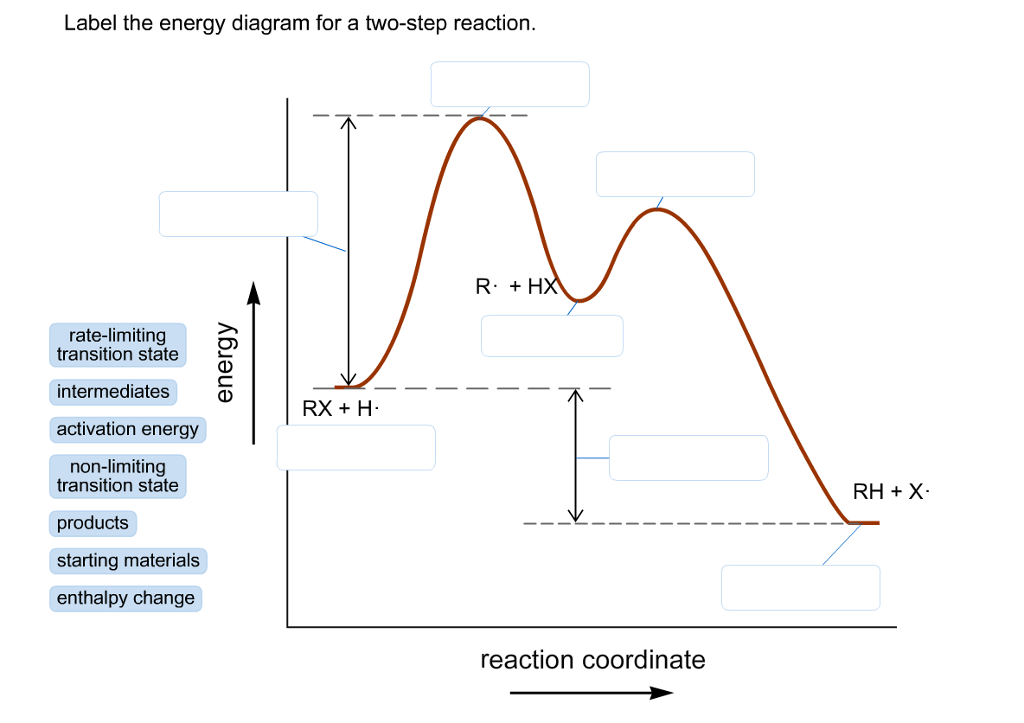
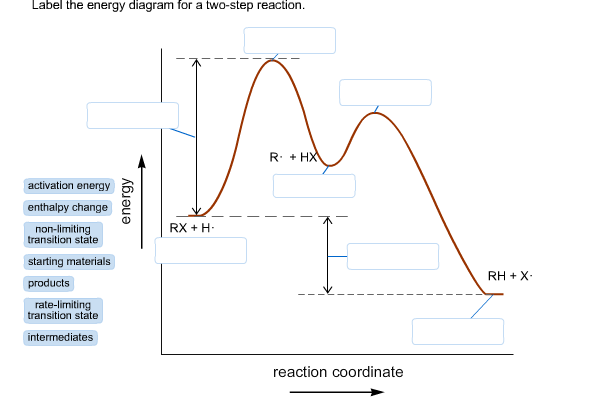
Label the energy diagram for a two-step reaction. Subject: Chemistry Price: 3.85 Bought 3 Share With. Label the energy diagram for a two-step reaction. enthalpy change transition state starting materials RX+H products rate-limiting transition state intermediates activation energy reaction coordinate
Draw a reaction energy diagram for a two-step reaction that has an endothermic first step and an exothermic second step. Label the reactants, transition states, reaction intermediate, activation energies, and enthalpy differences.
Label the energy diagram for a two step reaction. Heroin C21h23no5 Pubchem The activation energy for each step is labeled e a1 and e a2. Label the energy diagram for a two step reaction. Endothermic because energy is needed to break the a b bond. Each elementary step has its own activated complex labeled ac 1 and ac 2.

Draw An Energy Diagram For A Two Step Reaction Where The First Step Is Exothermic And The Reaction Overall Is Endothermic The First Step Is Rate Limiting Label Reactants Products And Intermediates
57CP. Draw an energy diagram for each reaction. Label the axes, the starting material, product, transition state, ΔH°, and E a. a. A concerted, exothermic reaction with a low energy of activation. b. A one-step endothermic reaction with a high energy of activation. c. A two-step reaction.
• An energy diagram for a two-step reaction with one intermediate. Energy Diagrams. 9 • Organic chemists use a technique called electron pushing, alternatively called arrow pushing, to depict the flow of electrons during a chemical reaction.

The Following Diagram Represents The Energy Profile Diagram For A Two Step Reaction Calculate The Ratio Of Change In Heat Energy Involved In Step I To Step Ii Img Src Https D10lpgp6xz60nq Cloudfront Net Physics Images Ps Chm Ix C06 E06 006 Q01
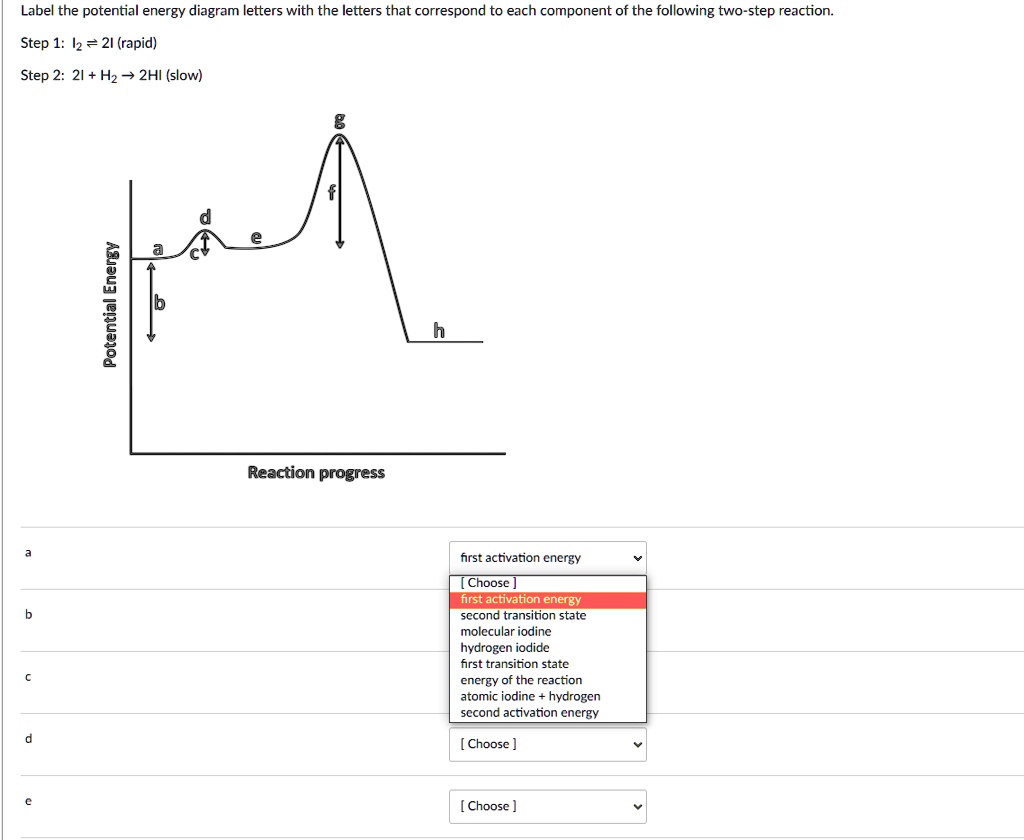
Solved Label The Potential Energy Diagram Letters With The Letters That Correspond To Each Component Of The Following Two Step Reaction Step 1 Iz 2i Rapid Step 2 21 Hz 2hi Slow

Question Video Identifying Steps In A Reaction Profile Diagram For A Two Step Chemical Reaction Nagwa













0 Response to "42 energy diagram for two step reaction"
Post a Comment