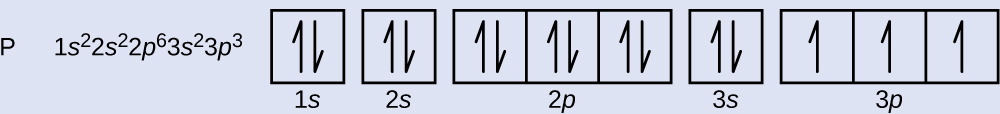
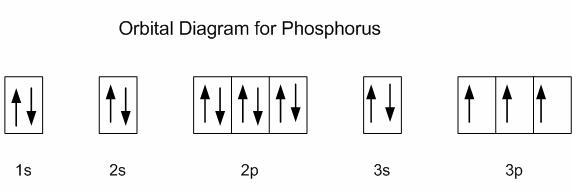
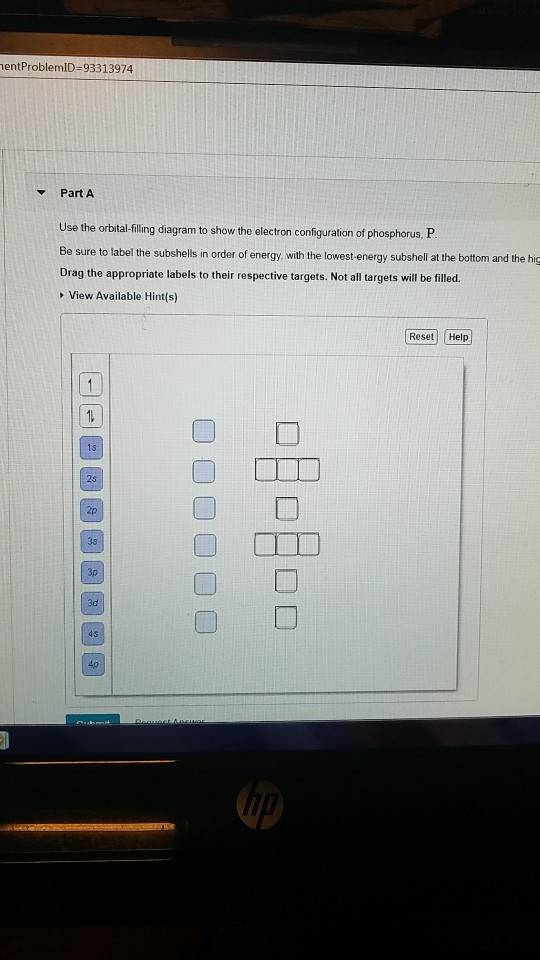
41 use the orbital-filling diagram to show the electron configuration of phosphorus, p.
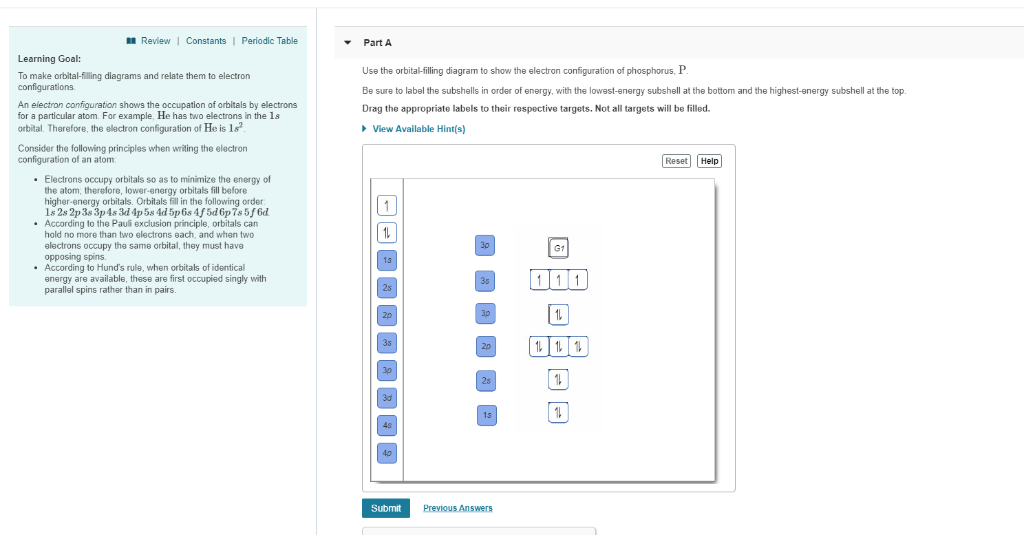
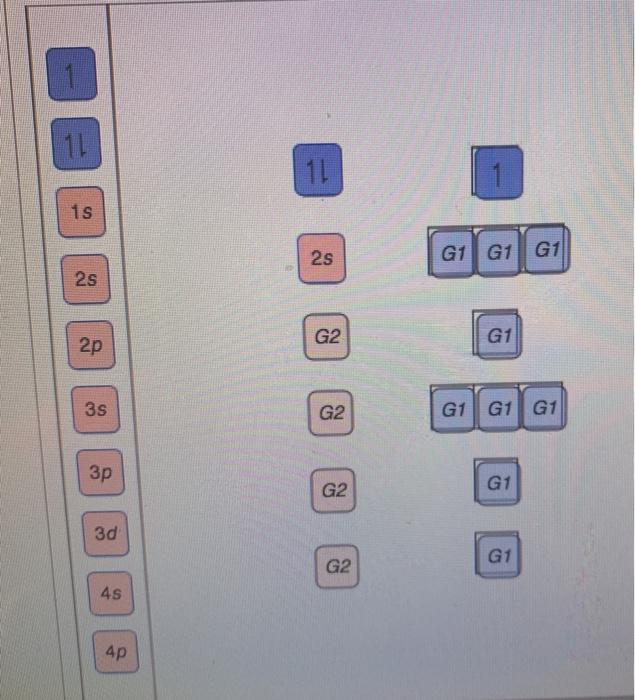
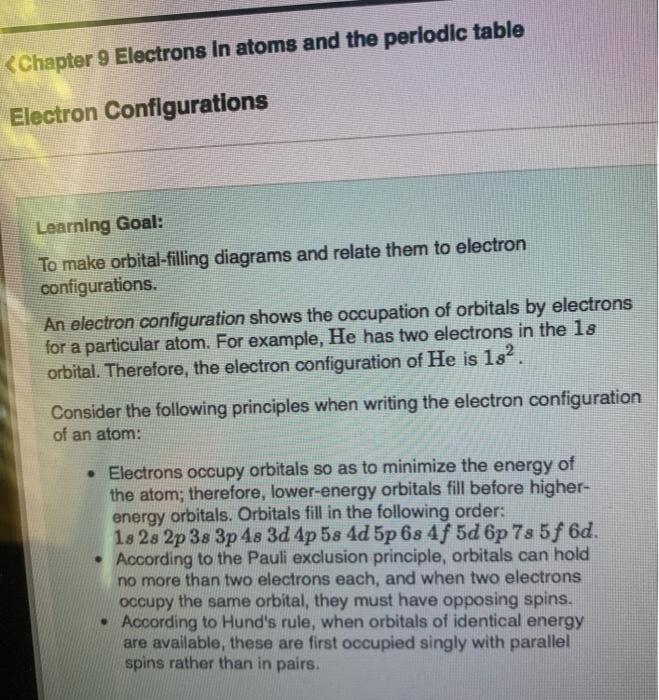
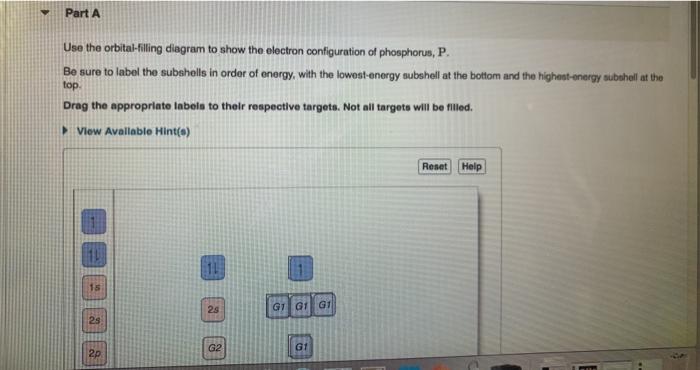
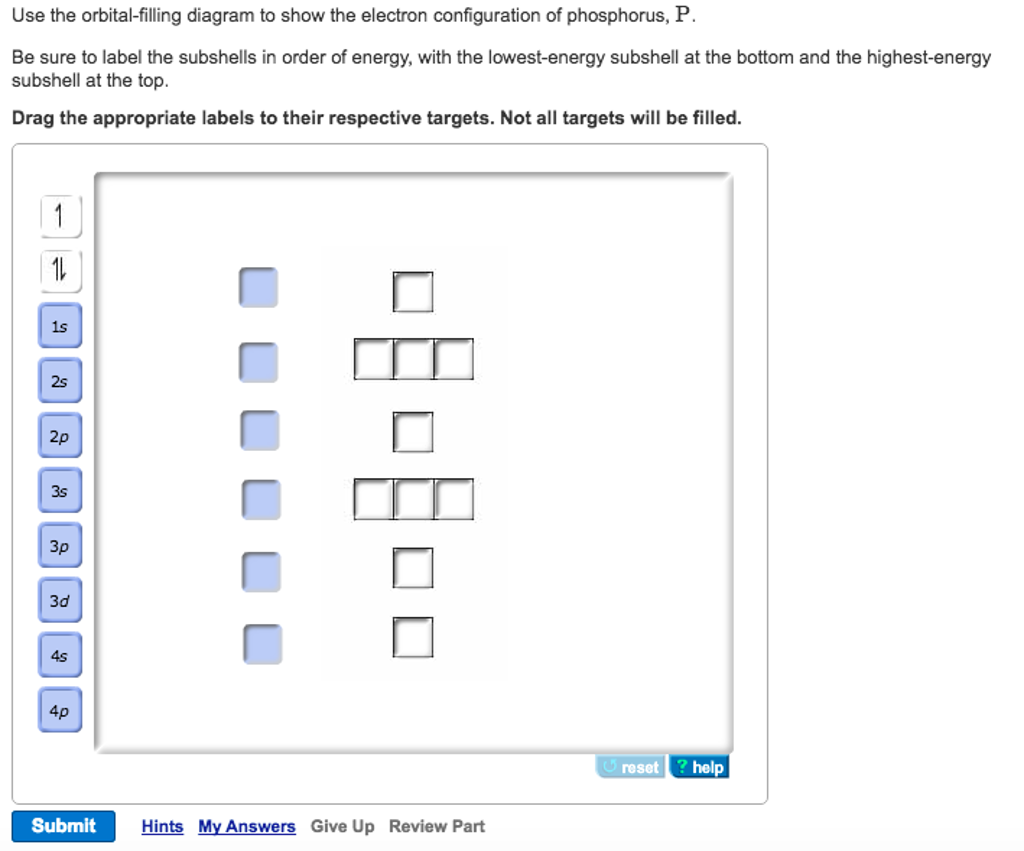
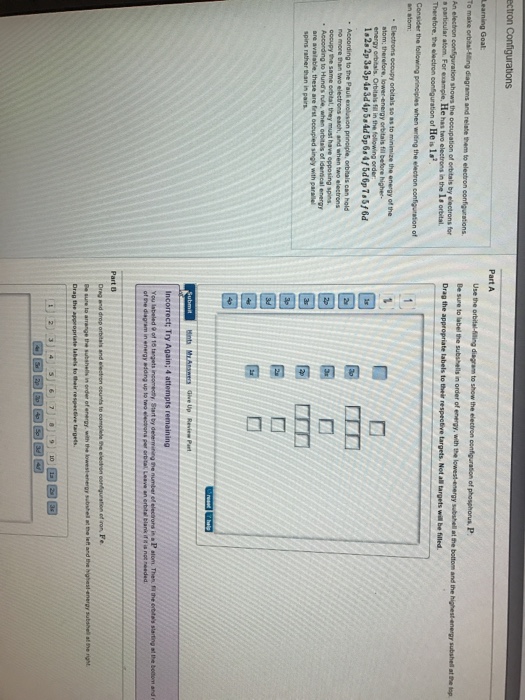
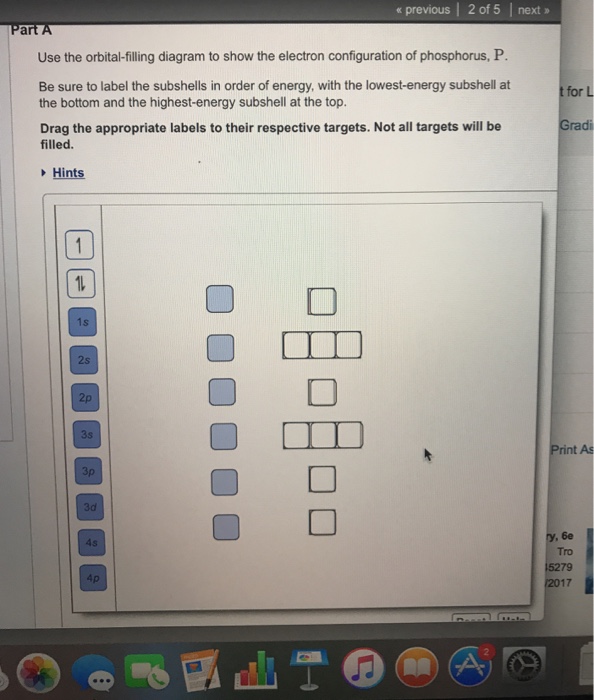
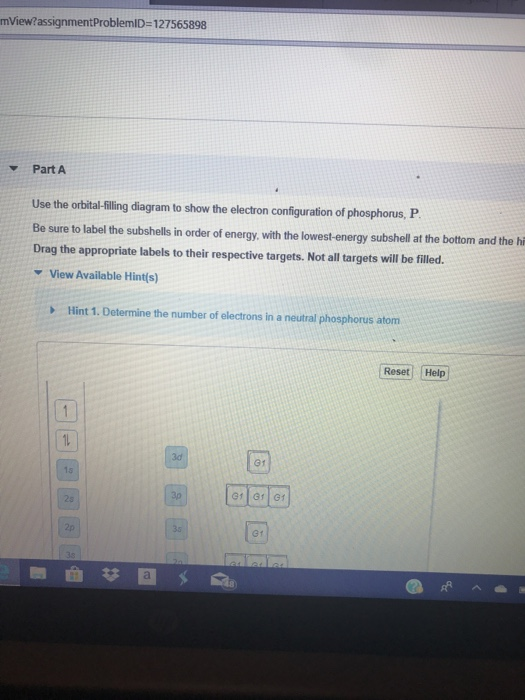
View?assignmentProblemID=127565898 Part A Use the orbital-filling diagram to show the electron configuration of phosphorus, P. Be sure to label the subshells in order of energy, with the lowest-energy subshell at the bottom and the hi Drag the appropriate labels to their respective targets. Not all targets will be filled.
Use the orbital-filling diagram to show the electron configuration of phosphorus P Orbital Diagrams: Electronic configuration is the distribution of electrons in the atomic orbitals like s,p,d,f,g....
30 May 2020 — The electron configuration of an atom indicates the number of valence ... We can clearly see that p orbitals are half-filled as there are ...
Use the orbital-filling diagram to show the electron configuration of phosphorus, p.
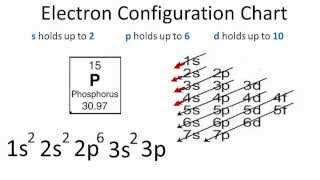
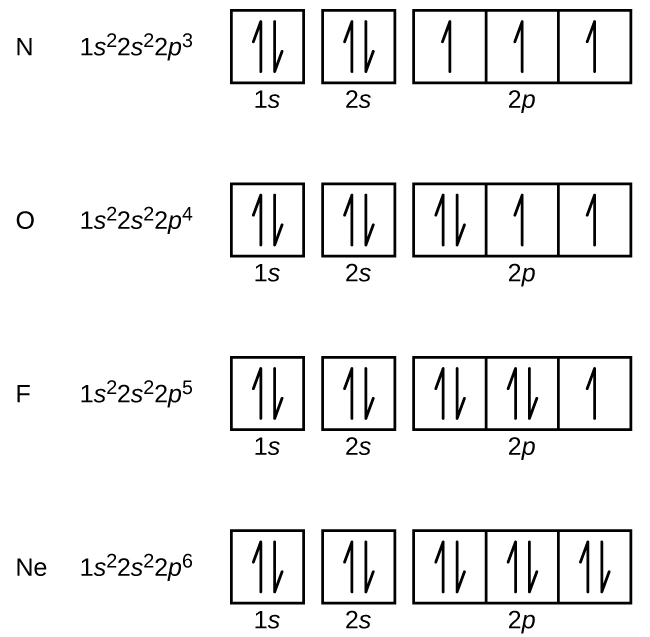
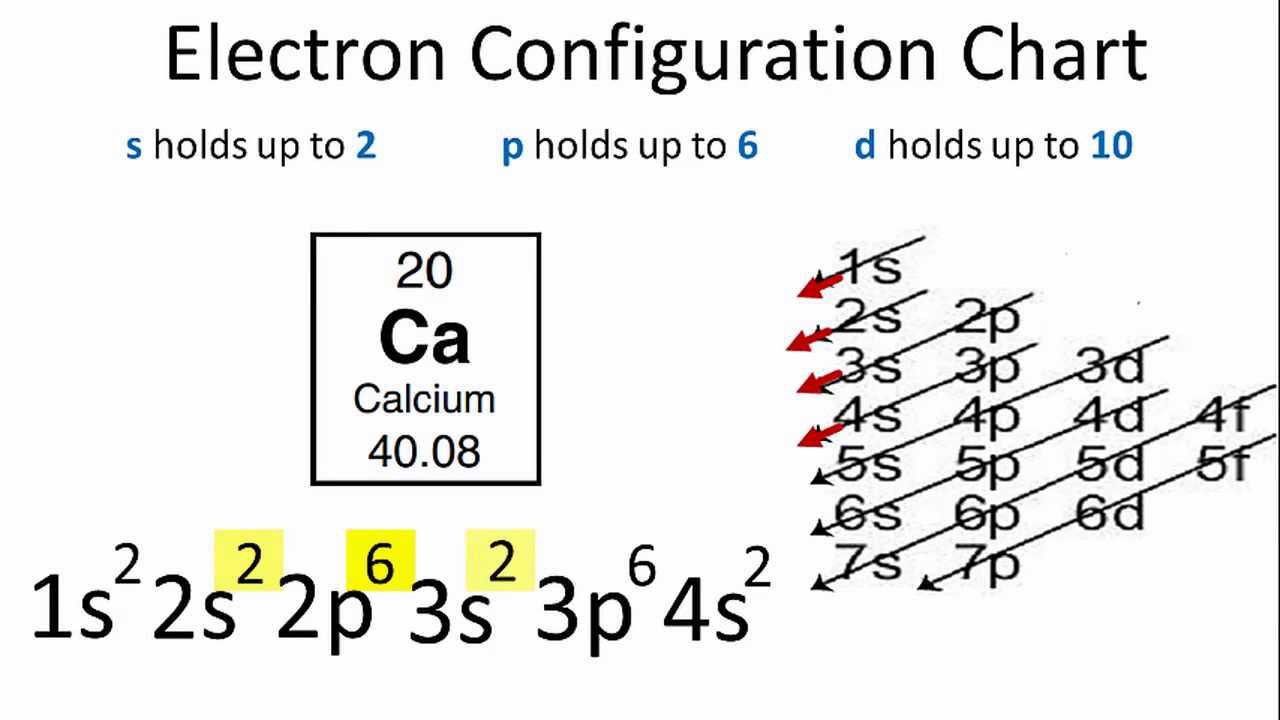
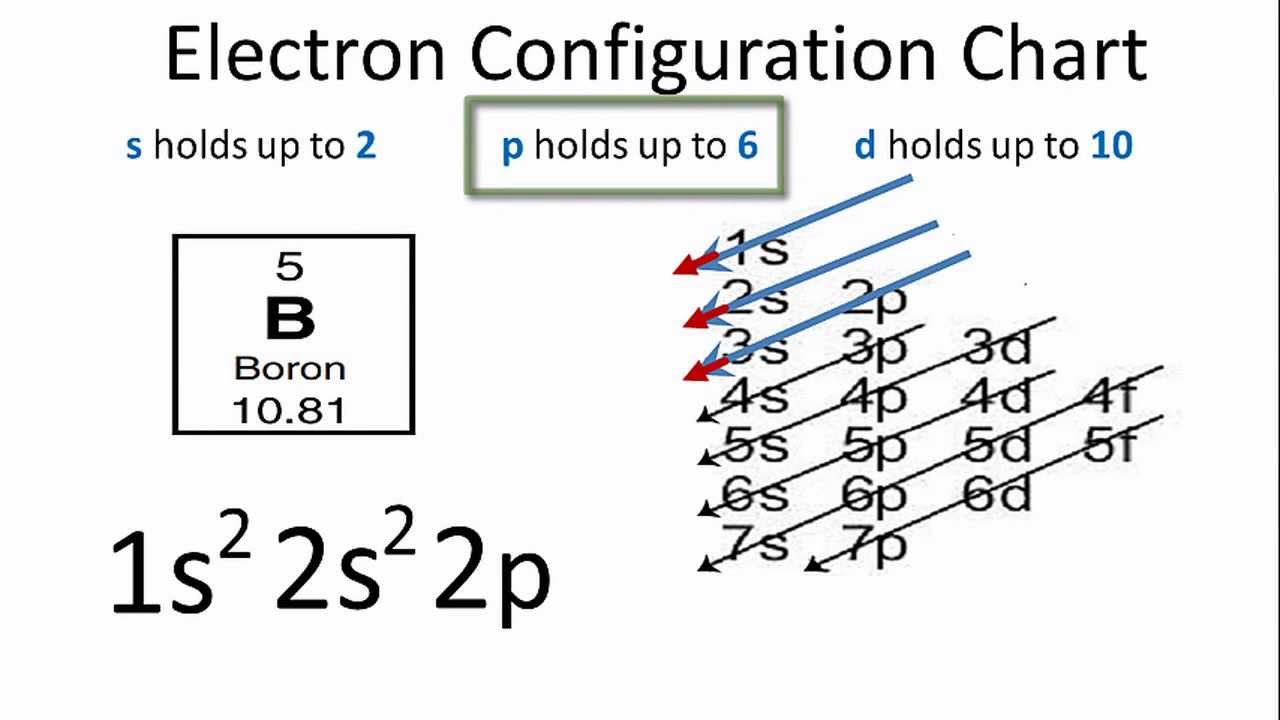
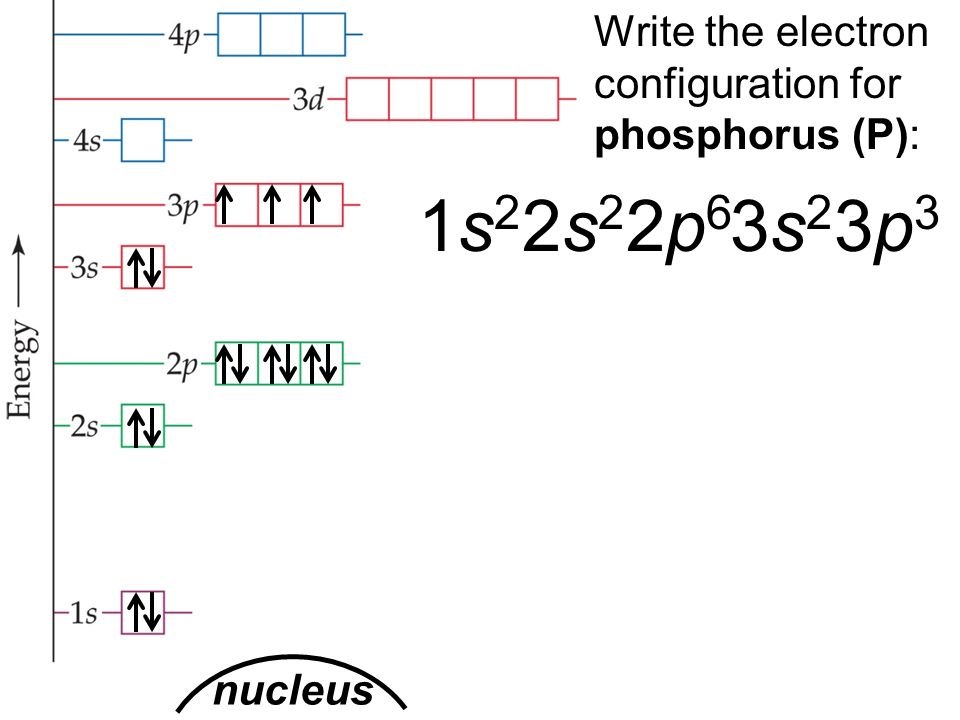
In writing the electron configuration for Phosphorus the first two electrons will go in the 1s orbital. Since 1s can only hold two electrons the next 2 electrons for Phosphorous go in the 2s orbital. The next six electrons will go in the 2p orbital. The p orbital can hold up to six electrons. We'll put six in the 2p orbital and then put the next two electrons in the 3s.
Use the orbital-filling diagram to show the electron configuration of phosphorus, P. Be sure to label the subshells in order of energy, with the lowest-energy subshell at the bottom and the highest-energy subshell at the Drag the appropriate labels to their respective targets. Not all targets will be filled.
Electron configurations are the summary of where the electrons are around a nucleus. As we learned earlier, each neutral atom has a number of electrons equal to its number of protons. What we will do now is place those electrons into an arrangement around the nucleus that indicates their energy and the shape of the orbital in which they are located. Here is a summary of the types of orbitals and how many electrons each can contain: So based on what we know about the quantum numbers and using the chart above, you need 2 electrons to fill an s orbital, 6 electrons to fill a p orbital, 10 electrons to fill a d orbital and 14 electrons to fill the f orbital. BUT what we haven”t discussed is how these orbitals get filled…the order of fill.
Use the orbital-filling diagram to show the electron configuration of phosphorus, p..
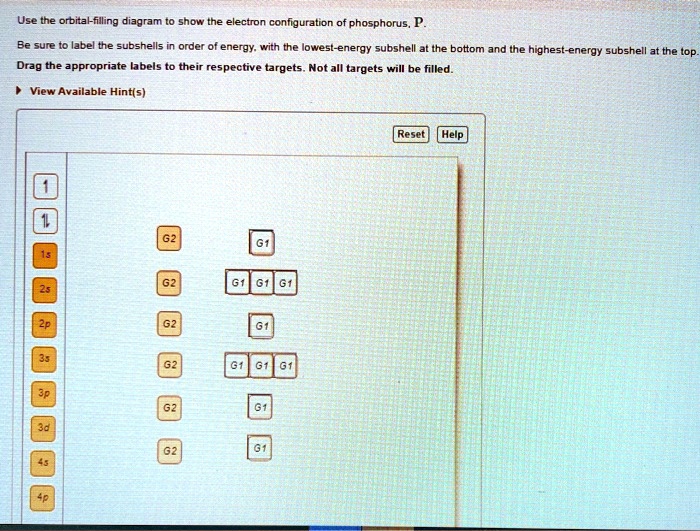
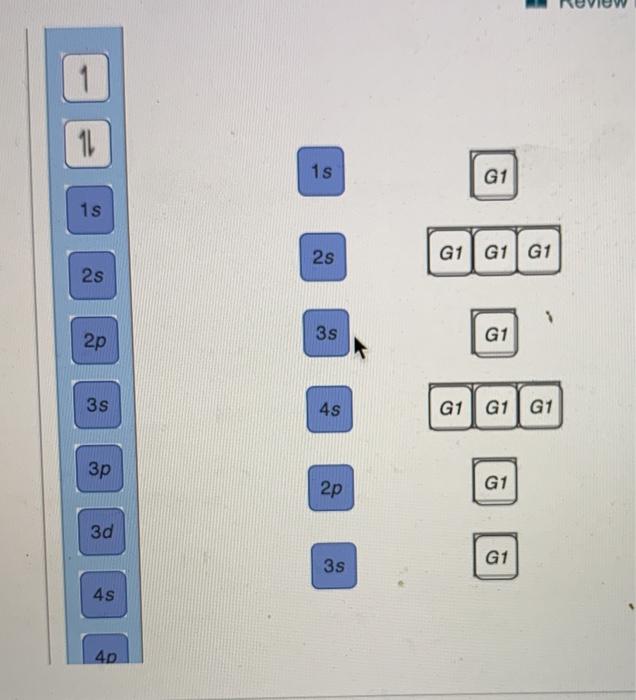
Transcribed image text: Use the orbital-filling diagram to show the electron configuration of phosphorus, P. Be sure to label the subshells in order of ...
21 Jan 2021 — Phosphorous is a chemical element that has an atomic number of 15. Its electron configuration with regards to electrons present in each shell is ...
Transcribed image text: Part A Use the orbital-filling diagram to show the electron configuration of phosphorus, P. Be sure to label the subshells in order ...
Transcribed image text: Use the orbital-filling diagram to show the electron configuration of phosphorus, P. Be sure to label the subshells in order of ...
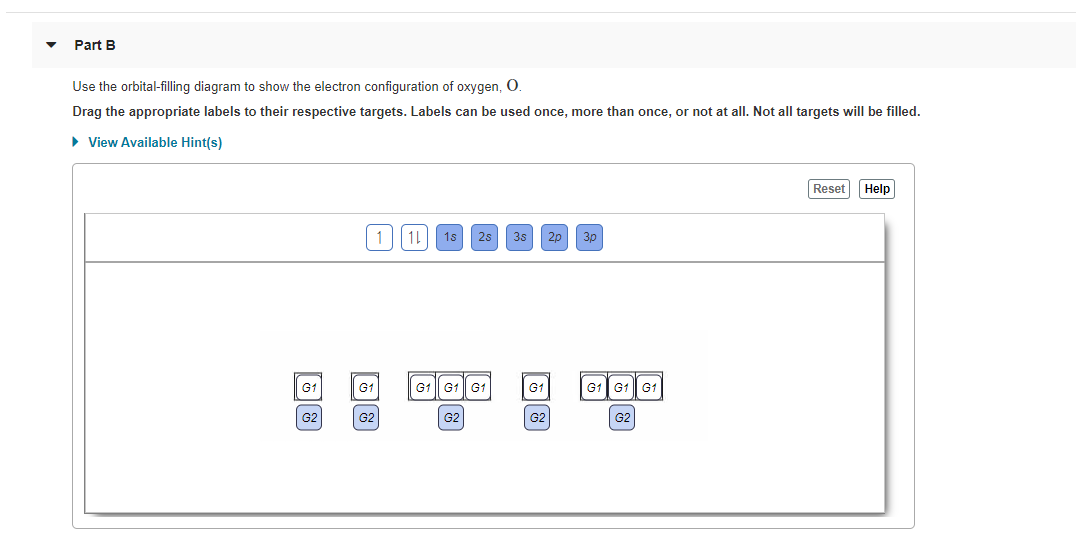
Use the orbital-filling diagram to show the electron configuration of helium, He Use the orbital-filling diagram to show the electron configuration of oxygen, O. Use the orbital-filling diagram to show the electron configuration of gallium, Ga.
Transcribed image text: Part A Use the orbital-filling diagram to show the electron configuration of phosphorus, P. Be sure to arrange the subshells in ...


















0 Response to "41 use the orbital-filling diagram to show the electron configuration of phosphorus, p."
Post a Comment